 | |
| Identifiers | |
|---|---|
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C23H33NO2 |
| Molar mass | 355.522 g·mol |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
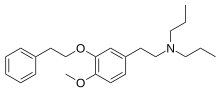
NE-100 or 4-methoxy-3-(2-phenylethoxy)-N,N-dipropylbenzeneethanamine is a selective sigma-1 receptor antagonist, with a reported binding affinity of Ki = 1.03 ± 0.01 nM, and more than 205 times selectivity over the sigma-2 receptor.
NE-100 was one of the earliest selective sigma-1 receptor ligands reported and has been widely used as a pharmacological tool. The original, eight step synthesis of NE-100 was reported by Atsuro Nakazato and colleagues of Taisho Pharmaceutical Company in 1999. More recently, Michael Kassiou and co-workers have reported a more expedient synthesis of NE-100 that proceeds in 56% unoptimized yield over 4 steps.
References
- Okuyama S, Nakazato A (1999). "NE-100: A novel sigma receptor antagonist". CNS Drug Rev. 2 (2): 226–237. doi:10.1111/j.1527-3458.1996.tb00299.x.
- Berardi F, Ferorelli S, Colabufo NA, Leopoldo M, Perrone R, Tortorella V (2001). "A multireceptorial binding reinvestigation on an extended class of sigma ligands: N- derivatives of 3,3-dimethylpiperidine reveal high affinities towards sigma1 and EBP sites". Bioorg. Med. Chem. 9 (5): 1325–35. doi:10.1016/s0968-0896(01)00011-6. PMID 11377189.
- Nakazato A, Ohta K, Sekiguchi Y, Okuyama S, Chaki S, Kawashima Y, Hatayama K (1999). "Design, Synthesis, Structure-Activity Relationships, and Biological Characterization of Novel Arylalkoxyphenylalkylamine s Ligands as Potential Antipsychotic Drugs". Journal of Medicinal Chemistry. 42 (6): 1076–1087. doi:10.1021/jm980212v. PMID 10090790.
- Banister SD, Moussa IA, Jorgensen WT, Chua SW, Kassiou M (2011). "Molecular hybridization of 4-azahexacyclododecane-3-ol with sigma (s) receptor ligands modulates off-target activity and subtype selectivity". Bioorg. Med. Chem. Lett. 21 (12): 3622–3626. doi:10.1016/j.bmcl.2011.04.098. PMID 21555222.